Hemodialysis
Baxter continues to push the boundaries of what’s possible in renal care. We are committed to advancing dialysis and hemodialysis treatment in ground-breaking and meaningful ways to give more choices to patients.
808,536
ESRD patients in the US1
84%
of incident patients are treated by In-Center Hemodialysis1
+3%
annual growth of prevalent ESRD cases in the US1
At Baxter, we don’t believe in delivering the same standardized therapy to all patients, but instead provide choices to select the therapy and treatment modality that is right for both patients and providers.
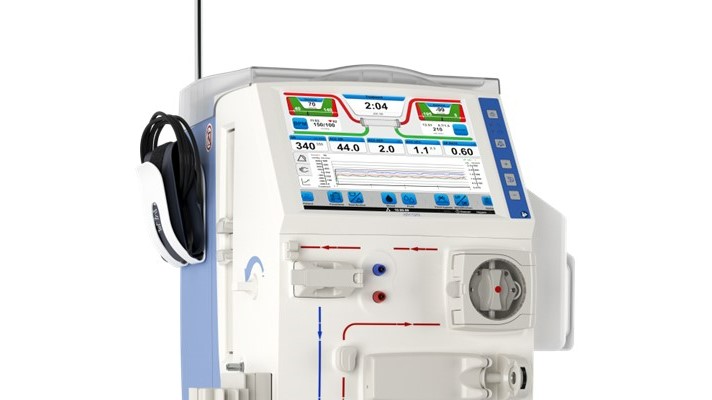
Our next-generation hemodialysis machine
Since first introduced in 1967, the Artificial Kidney platform has demonstrated a proven track record in dialysis care and significantly improved the field through groundbreaking technology and first-to-market advancements. The AK 98 Dialysis Machine continues this tradition and legacy.

The next evolution in dialysis - one step closer to the natural kidney 2
Accumulation of large-middle molecular uremic toxins contributes to disease burden in kidney failure patients, and high-flux membranes have limited ability to remove these large-middle molecules.2 HDx therapy enabled by Theranova dialyzers - equipped with MCO membrane - delivers superior removal of large-middle molecules compared to both conventional (high-flux) HD and HDF therapies, while maintaining stable serum albumin levels, and without changing your current HD machines and workflow.3,4
The AK 98 dialysis machine is intended to be used for intermittent hemodialysis and/or isolated ultrafiltration treatments of patients with chronic or acute renal failure or fluid overload upon prescription by a physician. The AK 98 dialysis machine is indicated to be used on patients with a body weight of 25 kg or more. The AK 98 dialysis machine is intended to be used by trained operators when prescribed by a physician, in a chronic care dialysis or hospital care environment. The AK 98 dialysis machine is not intended for Selfcare or Home use.
The Theranova Dialyzer is indicated for patients with chronic kidney failure who are prescribed intermittent hemodialysis. It provides an expanded solute removal profile with increased removal of various middle molecules (up to 45 kDa) that may play a pathologic role in the uremic clinical syndrome. The Theranova Dialyzer is not intended for hemofiltration or hemodiafiltration therapy. The total extracorporeal blood volume for the Theranova Dialyzer and the set should represent less than 10% of the patient's blood volume. For single use only.
Rx only. For the safe and proper use of these devices, refer to the Instructions for Use or Operator's Manual.
Baxter, AK 98, HDx, MCO and Theranova are trademarks of Baxter International Inc. or its subsidiaries.